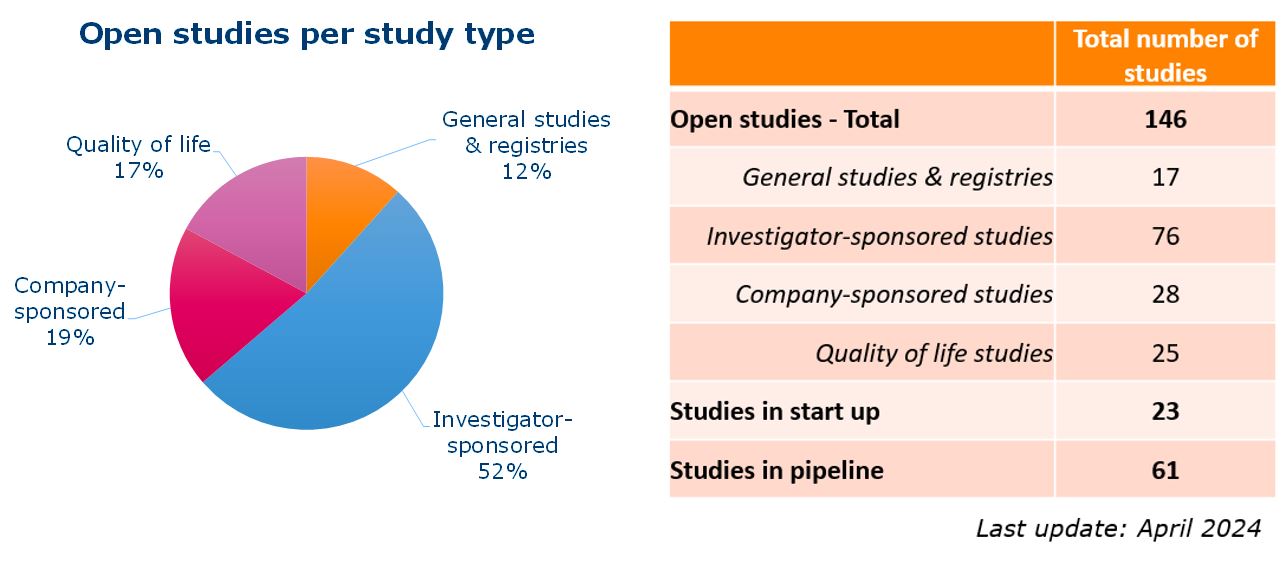
More than 100 clinical studies are ongoing at the Máxima center, and that number is constantly increasing. As the national center for pediatric oncology, the Máxima center coordinates all Dutch studies in the field of pediatric oncology. These include registration studies for new drugs of interest for pediatric oncology, such as brigatinib and venetoclax.
In addition, the Máxima center is more and more often acting as a sponsor of international studies. For example, Interfant-21 and CHIP-AML. Due to patient access and international collaborations (among others ITCC, Pedal, NANT, SIOPe and PNOC consortia), the Máxima center has a strong international position. There is no other comparable sponsor within the European Union.
The TDC is the central core facility (one-stop shop) for all clinical studies and data requests at the Máxima center.
The TDC handles the implementation and execution of all clinical studies for which patients are being recruited at the Máxima center. This includes studies initiated by researchers in academic centers as well as studies initiated by pharmaceutical companies. In addition, the TDC provides design and setup of investigator-initiated studies conducted both nationally and internationally.
This includes:
For this, the TDC provides:
The TDC is responsible for the processing, delivery and interpretation of data for the purpose of research. For clinical trials we work according to CDISC standards for the highest possible quality.
Our services include:
With some 100 motivated and enthusiastic employees, the TDC features broad expertise with which we strive to provide efficient and high-quality service. All employees undergo continuous training and much attention is paid to innovation.
The TDC works according to international standards (Good Clinical Practice) and regarding data according to the principles of FAIR (Findable, Accessible, Interoperable, Reusable). With our own quality management system and standard operating procedures (SOPs), we ensure quality. We play an active role in the integration of research and care and cooperate with international partners.
The Scientific Committee (SciCom), consisting of the Clinical Research Committee (CRC) and the Biobank and Data Access Committee (BDAC), reviews all research proposals involving patients of the Máxima center, their data and/or materials. (email to: scicom@prinsesmaximacentrum.nl)
The TDC is led by Prof. Dr. Michel Zwaan (pediatric oncologist) and Dr. Harm van Tinteren (statistician), supported by business operations manager Steven Vanhoutvin and team leaders Edith Schasfoort (site), Karolien Makkink (trial management) and Rinke Riezebos (data management).
The site team of the TDC handles the implementation of all studies for which patients are being recruited in the Máxima center. This concerns both studies initiated by investigators in academic centers as well as studies set up by pharmaceutical companies.
Trial management is concerned with the set-up and coordination of investigator-initiated studies conducted both locally and internationally by participating centers around the world. Trial managers work closely with investigators and provide end-to-end process management of clinical trials, supported by clinical trial assistants.
Depending on the type of study, trial management is responsible for:
Before start:
During the study:
Within the TDC, the Clinical Data Provisioning team is responsible for the processing and delivery of data for the purpose of research.
The team consists of several sub-teams, each with its own task:
Provide support in all phases of prospective clinical research, from design to final analysis
All clinical research is to be conducted in accordance with applicable laws and regulations, including ‘Good Clinical Practice’ (GCP). This is audited on a regular basis. The quality officers are responsible for the quality management system of the department. They also develop and teach the center-specific meeting of the BROK® course (Basic Course in Regulation and Organization for Clinical Researchers) for all staff involved in clinical research.
The Business Operations team is responsible for budgeting studies and drafting and completing all study-related contracts. They make arrangements and maintain contracts with pharmaceutical companies and other suppliers (e.g. software companies). For additional legal support, the TDC forwards your question to the Knowledge Transfer Office (KTO).